Chemical formula
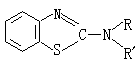

④The dithiocarbamates are super-fast accelerators, suitable for rapid vulcanization at room temperature, and also used as auxiliary accelerators.
In addition, there are substances that can improve the activity of organic accelerators, called vulcanization activators, that is, accelerators. The most widely used is zinc oxide, with an amount of 3 to 5 parts. The substance that prevents or delays the early vulcanization ("scorch") of the rubber compound during processing and parking before vulcanization is called a vulcanization retarder, that is, a scorch inhibitor. Scorch inhibitors with better effects include N-nitrosoaniline (anti-scorch agent NDPA), N-cyclohexyl sulfurized phthalimide (anti-scorch agent PVJ or CTP), etc. The amount of the former is 0.3 to 1 part, and the latter is 0.1 ~0.5 copies. The research work of anti-scorching agents is still very active.

product category
1. Rubber vulcanization aids, including vulcanizing agents (crosslinking agents), accelerators, activators and anti-scorching agents.
2. Rubber protective additives include antioxidants, antiozonants, anti-flex cracking agents, light stabilizers, ultraviolet light absorbers, harmful metal inhibitors, physical antioxidants, anti-termite agents, anti-rat bite agents, Biting agent, antifungal agent, etc.
3. Rubber reinforcing additives, including carbon black, white carbon black, metal oxides, inorganic salts, resins, etc.
4. Rubber adhesion additives, including m-methyl white system and cobalt salt system adhesives and adhesives
5. Process operation aids include peptizers, solubilizers, plasticizers, softeners, homogenizers, lubricants, dispersants, tackifiers, release agents, mold release agents, etc.
6. Special additives include colorants, foaming agents, defoamers, thickeners, creaming agents, wetting agents, emulsifiers, stabilizers, coagulants, heat-sensitive agents, anti-webbing agents, preservatives, preservatives , Flame retardants, antistatic agents, fragrances and anti-biting agents, etc.



 English
English Português
Português Español
Español русский
русский 中文简体
中文简体










